In case of positive results from these trials, rheumatologists will become more confident about Krystexxa, thereby enabling them to prescribe Krystexxa to more patients in their care. The MIRROR double-blind placebo-controlled registrational, also evaluating the co-administration of Krystexxa with methotrexate in uncontrolled gout patients, commenced in June 2019. In this trial, the company administered Krystexxa and methotrexate to 14 patients. The company expects data from the MIRROR open-label study which started in the third quarter of 2018, in early 2020. Here, the company is assessing if methotrexate, an immunomodulator used commonly by rheumatologists can help dampen the immune response to Krystexxa and thereby increase the response rate to Krystexxa. Horizon is currently studying the effect on the response rate of Krystexxa with the administration of methotrexate in patients with uncontrolled gout in the two MIRROR immunomodulation trials. Krystexxa is also being studied in additional indications Other oral urate-lowering therapies focus on the overproduction or under excretion of uric acid. While only 10% of uric acid filtered through kidneys can be excreted, the renal excretion of allantoin is almost 10 times better than uric acid. Unlike other drugs, Krystexxa converts the source of uric acid crystals, urate, into a water-soluble substance, allantoin. The drug has a unique mechanism of action in reversing disease progression in uncontrolled gout. It has been seen that gout patients have an average of four comorbidities, with the most common ones being hypertension, hyperlipidemia, chronic kidney disease, coronary artery disease, and diabetes Mellitus. Patients with high uric acid levels generally have multiple comorbidities. 
Here, uric acid deposits in bones and joints as well as other organs such as heart and kidney. Gout is a common type of inflammatory arthritis that is characterized by higher serum uric acid levels. Horizon now expects 25% YoY revenue growth for the drug in 2019. In 2018, the drug reported $258.9 million in net sales, a YoY rise of 65%. The addressable patient population is 100K. 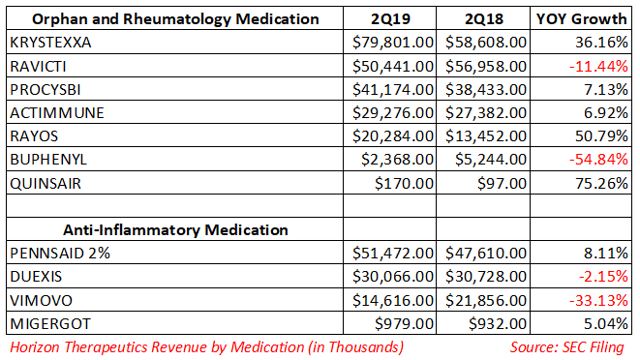
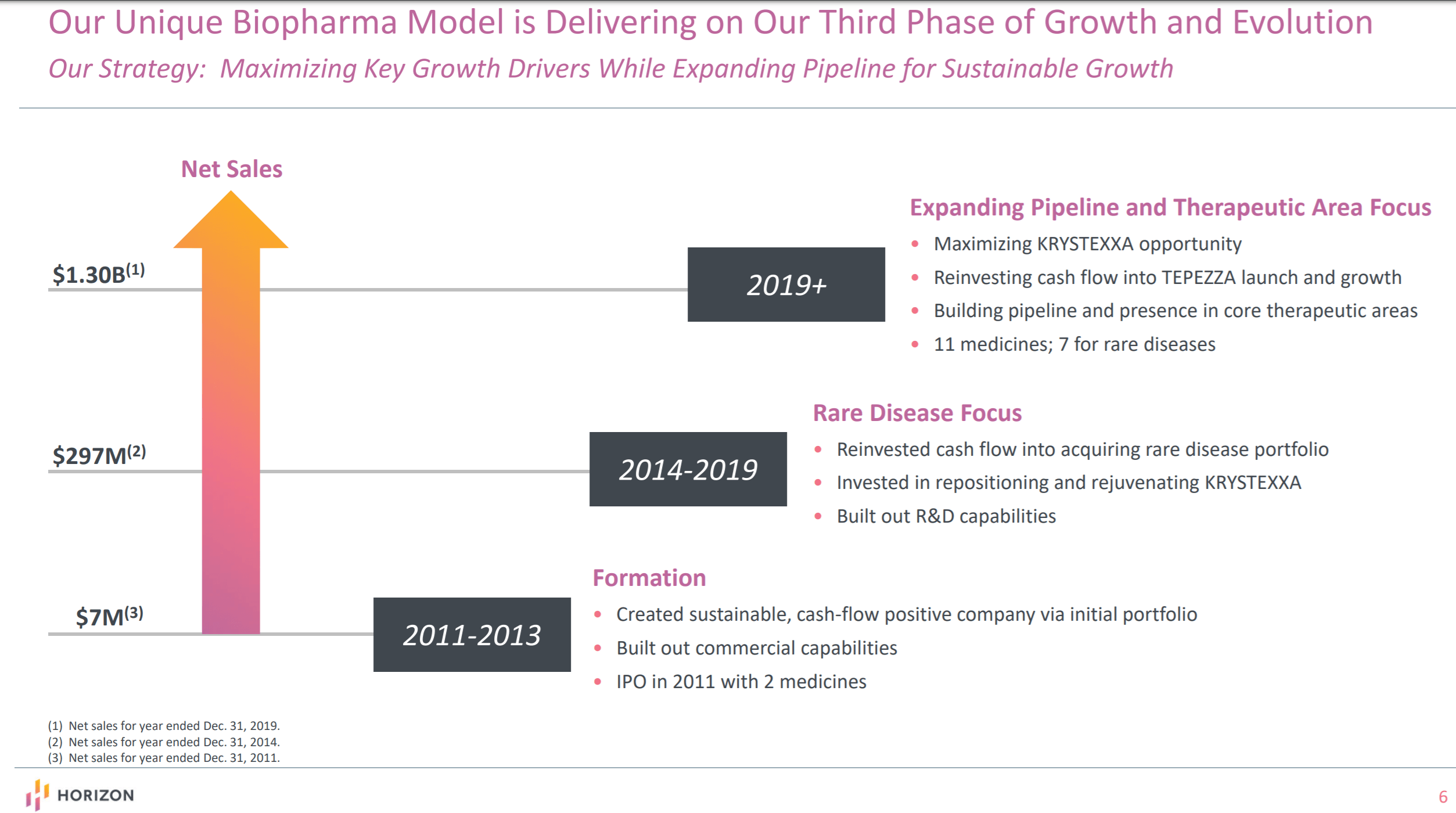
annual peak sales for Krystexxa in excess of $750 million. Horizon Therapeutics transformed its growth trajectory and almost quadrupled its sales in less than four years. When Horizon Therapeutics first acquired the first and only FDA approved therapy for chronic refractory gout, Krystexxa, from Crealta Holdings, it was an underperforming and undervalued asset. Krystexxa has been a phenomenal success story for Horizon Therapeutics The company's lead investigational asset, teprotumumab, is also headed for FDA approval in TED (thyroid eye disease) indication. Besides, the company is also strengthening its rare disease pipeline through internal development, licensing, and acquisitions. Horizon Therapeutics is focused on maximizing the value of Krystexxa. 
Krystexxa, Ravicti, and Procysbi are the company's major revenue drivers. The company currently markets approximately 10 drugs, of which 6 are for rare and immunological conditions. Once known mainly for its primary care drugs, the company has emerged as a leading rare disease biopharmaceutical company. Today, Horizon Therapeutics stands at the cusp of a major transformation. Today, we will be studying why Horizon Therapeutics ( NASDAQ: HZNP) is an attractive investment for 2020. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
